The Heterocycle Isostere Explorer: A Computational Tool for the Discovery of Novel Aromatic Heterocyclic Isosteres

University of Oxford, Exscientia Plc, University of Liverpool
https://doi.org/10.1021/acs.jmedchem.5c03118
Aromatic heterocycles are among the most common groups in drug discovery, with over 60% of approved drugs containing one of these groups. Due to their prevalence, heterocycle isosteres are frequently used in drug development to improve the potency or modify the properties of these molecules. While libraries of these isosteres exist, most are based on previously made compounds and are unable to suggest novel isosteres. In this work, the researchers have developed the Heterocycle Isostere Explorer (HCIE), a Python-based tool which uses vector-based ligand alignment to search isosteres in a custom-built library of aromatic heterocycles. This tool was then used to propose novel isosteres for aromatic heterocycles used in drug discovery.
Do Amino-Oxetanes Resemble Amides? A Matched Molecular Pairs Property and Structural Comparison

Imperial College London, Pfizer
https://doi.org/10.1021/acs.jmedchem.5c02614
Amides are another group commonly found in drug-like molecules. However, they can display poor plasma stability due to hydrolysis of the amide bond. Amino oxetanes have previously been suggested as bioisosteres of this group. In this study, the authors synthesised a series of arylamino-oxetane and benzamide matched molecular pairs and compared their properties. They found that amides and amino oxetanes have similar ADME properties, although their confirmation is more closely matched to sulphonamides.
Amide-to-Chloroalkene Substitution for Peptide Backbone Modification to Enhance Membrane Permeability

Shizuoka University, Hokkaido University, University of Tsukuba
https://doi.org/10.1021/acs.jmedchem.5c02090
Peptide-based drug discovery and therapeutics are rapidly growing due to their generally higher potency, selectivity, and lower toxicity. However, one key drawback of peptide-like drugs is their poor membrane permeability, due to the highly polar amide bonds which make up the peptide backbone. Many methods have been explored to get around this issue, such as N-methylation and ester and thioamide replacements of amide bonds. Chloroalkene replacements of amides have previously been used to improve the proteolytic stability of peptides, but their effects on permeability have been underexplored. This paper assesses their impact on permeability and shows that the chloroalkene provides the largest improvement in peptide permeability when compared to other common replacements.
The Affinity Advantage

Mark A. Murcko - Disruptive Biomedical
https://doi.org/10.1021/acs.jmedchem.5c03222
In recent years, it has been argued that potency is an almost trivial part of drug discovery, as many potent compounds have been discovered, but lack acceptable ADMET or DMPK profiles to be taken further forward in development. In this perspective, the author argues that potency has been undervalued, with not enough effort put into optimising this property. He lists several advantages, responses to objections, and practical tips for gaining an “affinity advantage”.
Orally Bioavailable Cyclin A/B RxL Inhibitors: Optimization of a Novel Class of Macrocyclic Peptides That Target E2F-High and G1–S-Checkpoint-Compromised Cancers
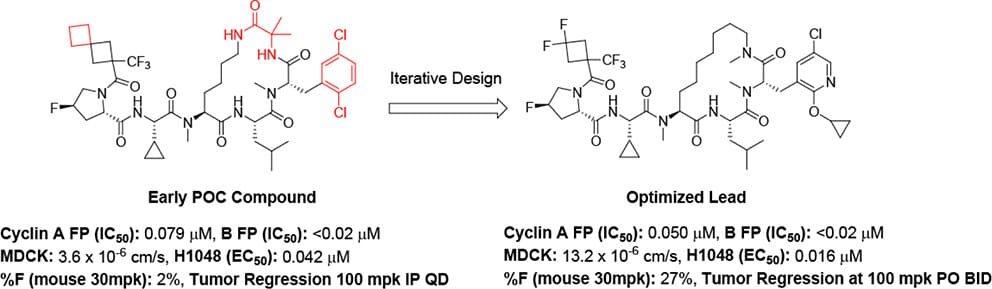
Circle Pharma
https://doi.org/10.1021/acs.jmedchem.5c02445
Macrocyclic peptides are a common modality for targeting protein-protein interactions (PPIs) due to their ability to mimic the target’s natural substrate. One challenge with macrocyclic peptides is their generally poor cell permeability and oral bioavailability. This is a problem when trying to target intracellular PPIs with oral dosing. This work describes Circle Pharma’s journey of converting their early proof of concept cyclin A/B RxL inhibitors in to orally bioavailable drugs, which are now being evaluated in phase I.
High-throughput ligand diversification to discover chemical inducers of proximity

The Scripps Research Institute, CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences, Dana-Farber Cancer Institute, Centre for Targeted Protein Degradation, Albert Einstein College of Medicine
https://doi.org/10.1038/s41589-025-02137-2
PROTACs and molecular glues are two examples of chemical inducers of proximity, which represent new modalities in drug discovery. While rational design of PROTACs has been achieved, the discovery of new molecular glues remains limited, mainly relying on serendipity. In this study, high-throughput chemistry and miniaturised cell-based screens were used to identify new molecular glues for ENL (eleven-nineteen leukaemia) and BRD4 (bromodomain-containing protein 4).
From Serendipity to Strategy: Rationalizing Molecular Glue Discovery and Proximity-Induced Pharmacology through Chemical Biology

Imperial College London
https://doi.org/10.1021/jacs.5c12299
Another article highlights that molecular glue discovery has primarily relied on serendipity to drive discoveries. The authors describe how advances in library design and screening technologies are allowing for more rational design of molecular glues.
Direct-to-Biology: Streamlining the Path From Chemistry to Biology in Drug Discovery

Johannes Gutenberg-University
https://doi.org/10.1002/cmdc.202501080
Direct-to-biology (D2B) is a screening technique that uses high-throughput synthesis and screening of crude reactions to accelerate DMTA (design-make-test-analyse) cycles. This review article highlights and summarises publications in this area.
Honorable mentions
Analyses of Recent Hit-Finding Campaigns for Difficult Targets Provides Guidance for Informed Integrated Hit Discovery
https://doi.org/10.1021/acsmedchemlett.5c00676
Misconceptions in Unbound Volume of Distribution and Their Implications for Pharmacokinetic Scaling and Drug Design
https://doi.org/10.1021/acsmedchemlett.6c00063
Discovery of BMS-986458, a Potent and Selective B-Cell Lymphoma 6 Protein Ligand-Directed Degrader, for the Treatment of B-Cell Non-Hodgkin Lymphoma
https://doi.org/10.1021/acs.jmedchem.5c03123
Discovery of EGT710, an Oral Nonpeptidomimetic Reversible Covalent SARS-CoV‑2 Main Protease Inhibitor
https://doi.org/10.1021/acs.jmedchem.5c02360